Batteries Get a Boost from ‘Pickled’ Electrolytes
Computer Simulations Reveal How Additive Improves Li-On Battery Performance
August 1, 2018
By Margie Wylie
Contact: cscomms@lbl.gov
Battery researchers at the U.S. Department of Energy’s (DOE) Argonne National Laboratory have used computer simulations to help reveal the mechanism behind a common additive known to extend the life of lithium-ion batteries. At its heart is a chemical reaction similar to pickling.
The energy storage system of choice for electric vehicles is the lithium-ion battery, but the cathode in these batteries limits their maximum energy storage capacity. Researchers have identified high-capacity cathode materials that operate at high voltage, but their long-term use remains problematic. As the cell charges and discharges, the materials react with the liquid electrolyte degrading it and stalling performance.
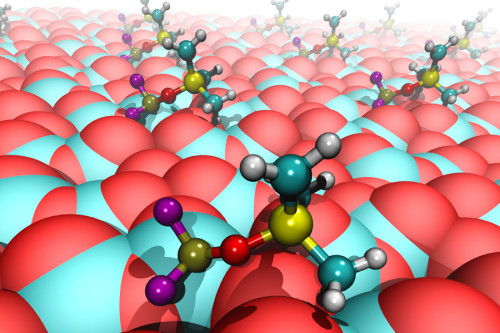
This illustration shows pickled electrolyte molecules (PF2OSiMe3) binding to reaction centers on a Li-On battery’s cathode surface where it protects the cathode from degrading and extends battery life. (Image by Argonne National Laboratory / Juan C. Garcia.)
Mixing a performance-boosting additive into the liquid electrolyte—in this case, trimethylsilyl phosphite, or TMSPi — can stall decomposition by adding a protective layer to the cathode surface. Until now, however, scientists didn’t understand how TMSPi worked.
Using a series of simulations carried out at Lawrence Berkeley National Laboratory’s (Berkeley Lab) National Energy Research Scientific Computing Center (NERSC), ANL researchers found that TMSPi itself isn’t directly involved in protecting the cathode. Instead a chemical derivative (PF2OSiMe3) does the job.
One among many such products, PF2OSiMe3 slowly forms as the lithium salt in the electrolyte reacts with TMSPi. Argonne Senior Materials Scientist Daniel Abraham compared the process to anaerobic fermentation of cucumbers in brine, “which gives us tasty pickles.”
Compute-Intensive Process
Uncovering this “pickling” mechanism was a computationally intensive process, said Hakim Iddir, an Argonne physicist and NERSC P.I. for the project. “The supercell [simulation box] needed to accommodate large molecules interacting with the surface of the cathode material had to be very large, around 400 atoms,” Iddir said. “We had to do two-step, high-level DFT calculations to achieve the necessary electronic convergence using several different configurations of molecules and surfaces,” he said.
A typical set of calculations (executed on NERSC's Cori supercomputer) used 288 processing cores and ran for over 24 hours. With so many different configurations to test, the researchers did around 60 simulations requiring a total of about 41,500 computing hours, according to Iddir.
“NERSC provided the resources – the number of cores, the software, stability and performance – that we needed to conduct such a demanding project,” said Juan C. Garcia, the Argonne postdoctoral fellow who ran the calculations. NERSC is a DOE Office of Science User Facility.
Proof in the Pickling
Their calculations found that PF2OSiMe3 strongly binds to reaction centers on the cathode surface without causing the detrimental removal of oxygen from the surface. This surface-bound molecule can further react with the electrolyte, transforming into a still-stronger binding molecule that permanently caps the reaction centers on the cathode, stabilizing the interface between liquid electrolyte and solid electrode. “As a result,” Abraham reports, “battery performance actually improves as the TMPSi electrolyte additive ages.”
Abraham also reports that this early-stage research study has an important practical application. “Now that we better understand the mechanism for the cathode-protective action by the phosphite, we can be more systematic in finding new ways of achieving and improving this pickling of the electrolyte additive.”
“Chemical ‘Pickling’ of Phosphate Additives Mitigates Impedance Rise in Li Ion Batteries,” published in The Journal of Physical Chemistry, describes the work.
The DOE’s Vehicle Technologies Program, within its Office of Energy Efficiency and Renewable Energy, funded the work under the multi-institutional Deep Dive entitled “Enabling High-Energy/High Voltage Lithium-Ion Cells for Transportation Applications.” In addition to NERSC, the team also used high-performance computing facilities of the Laboratory Computing Resource Center at Argonne.
This story was adapted from an Argonne National Laboratory press release.
About NERSC and Berkeley Lab
The National Energy Research Scientific Computing Center (NERSC) is a U.S. Department of Energy Office of Science User Facility that serves as the primary high performance computing center for scientific research sponsored by the Office of Science. Located at Lawrence Berkeley National Laboratory, NERSC serves almost 10,000 scientists at national laboratories and universities researching a wide range of problems in climate, fusion energy, materials science, physics, chemistry, computational biology, and other disciplines. Berkeley Lab is a DOE national laboratory located in Berkeley, California. It conducts unclassified scientific research and is managed by the University of California for the U.S. Department of Energy. »Learn more about computing sciences at Berkeley Lab.







