Theoretical Study on Catalysis by Protein Enzymes, Ribosome, and Molecular Motors
NERSC Annual Report 2001
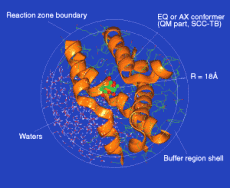
The model protein-substrate system used in the QM/MM molecular dynamics simulation of chorismate mutase (CM) based on the X-ray structure of yeast CM. During the molecular dynamics simulation, it was found that the inactive conformers were rapidly converted into the reactive chair conformation through the motion of active site residues; such a conversion was not observed in solution. Thus the results highlight the importance of enzyme dynamics in catalysis.
Research Objectives
This project's goal is to develop a greater understanding of the mechanisms involved in enzyme catalysis and related protein functions. We are studying the protein enzymes chorismate mutase, flavoxireductase, and aminopeptidase, and a nucleic acid enzyme, the hammerhead ribosome. We are also studying another class of enzymes known as molecular motors, which play important roles in bioenergy transduction and gene replication.
Computational Approach
For active-site models in the gas phase, Gaussian98 and NWChem are used forab initio or density functional calculations. To determine the catalytic mechanism in the presence of the enzyme environment, the CHARMM program (developed by the Karplus group) is used for a combined quantum and molecular mechanics (QM/MM) approach.
Accomplishments
We studied the role of tunneling for two proton transfer steps in reactions catalyzed by triosephosphate isomerase (TIM). The effect of tunneling on the reaction rate is less than a factor of 10 at room temperature; the tunneling became more important at lower temperatures. The imaginary frequency mode and modes having large contributions to the reaction path curvature were localized on the atoms in the active site, within 4 Å of the substrate. This suggests that only a small number of atoms close to the substrate and their motions directly determine the magnitude of tunneling.
In horse liver alcohol dehydrogenase (LADH) proton and hydride transfers, proton transfers proceeded in a virtually concerted fashion before hydride transfers. The catalytic efficiency of LADH was low for a pH below 5.5, and the hydride transfer was hardly affected for a pH between 5.5 and 8.1. Perturbation analysis of the QM/MM energies suggests a number of charged residues close to the active site as well as the phosphate groups in NAD+ make important contributions to the energetics of proton and hydride transfer reactions.
Chorismate mutase (CM) acts at the first branch-point of aromatic amino acid biosynthesis and catalyzes the conversion of chorismate to prephenate. Two nonreactive conformers of chorismate were found to be more stable than the reactive pseudo-diaxial chair conformer in solution. When these inactive conformers were bound to the active site, they rapidly converted to the reactive chair conformer. This suggests that the enzyme binds the more prevalent nonreactive conformers and transforms them into the active form in a step prior to the chemical reaction.
Significance
Despite the growing availability of enzyme crystal structures, details of the chemical mechanisms employed by enzymes to achieve their catalytic efficiency remain elusive. This is mainly because the chemical events of bond formation and cleavage that define the reaction are exceedingly short and currently inaccessible to direct experimental measurement. It is also very difficult to probe directly the coupling between chemical events and conformational transitions with atomic details, which remains a major obstacle for understanding the working mechanism of molecular motors. Theoretical studies, therefore, are of great value for providing insights into these mechanisms.
Publications
H. Guo, Q. Cui, W. N. Lipscomb, and M. Karplus, "Substrate conformational transitions in the active site of chorismate mutase: Their role in the catalytic mechanism," Proc. Natl. Acad. Sci. USA98, 9032 (2001).
Q. Cui and M. Karplus, "Triosephosphate isomerase (TIM): A theoretical comparison of alternative pathways," J. Am. Chem. Soc.123, 2284 (2001).
P. D. Lyne and M. Karplus, "Determination of the pKa of the 2'-hydroxyl group of a phosphorylated ribose: Implications for the mechanism of hammerhead ribozyme catalysis," J. Am. Chem. Soc. 122, 166 (2000).
About NERSC and Berkeley Lab
The National Energy Research Scientific Computing Center (NERSC) is a U.S. Department of Energy Office of Science User Facility that serves as the primary high performance computing center for scientific research sponsored by the Office of Science. Located at Lawrence Berkeley National Laboratory, NERSC serves almost 10,000 scientists at national laboratories and universities researching a wide range of problems in climate, fusion energy, materials science, physics, chemistry, computational biology, and other disciplines. Berkeley Lab is a DOE national laboratory located in Berkeley, California. It conducts unclassified scientific research and is managed by the University of California for the U.S. Department of Energy. »Learn more about computing sciences at Berkeley Lab.







