Learning From Photosynthesis to Create Electricity
Researchers use NERSC supercomputers to investigate photosynthetic materials
May 15, 2012
NERSC Contact: Linda Vu, cscomms@lbl.gov
TACC Contact: Adam Dubrow, aarondubrow@tacc.utexas.edu
Solar power could transform the energy landscape in the United States, reducing the nation's reliance on coal and natural gas for electricity. Today, however, solar power remains more expensive on average than fossil fuels.
"You may think that the sun is abundant, but traditional photovoltaics require rare-earth elements, and a lot of them are imported from areas that have wars or where it is difficult to extract, which raises the cost," says Margaret Cheung, Assistant Professor of Physics at the University of Houston.
She notes that if we learn from plants, which use only common elements—hydrogen, nitrogen, carbon, oxygen and some others—to convert sunlight into energy, then we’ll be able to bring down the cost of solar power. This is why researchers are looking at bio-inspired materials as possible resources for solar energy.
In the 1990s, an Arizona State University research group led by chemistry professor Devens Gust made a huge advance in this field by creating the carotenoid-porphyrin-C60 molecular triad, a novel material that converts sunlight into chemical energy by mimicking photosynthesis. However, the material has been difficult to commercialize because it can only be controlled, or confined, in experimental labs.
Using 2 million computer hours at the Department of Energy’s National Energy Research Scientific Computing Center (NERSC) and 2.5 million computer hours at the Texas Advanced Computing Center (TACC), Cheung and her team explored the role that confinement, temperature, and solvents play in the stability and energy efficiency of the light-harvesting triad. Their results provide a way to test, tailor, and engineer nano-capsules with embedded triads that, when combined in large numbers, could greatly increase the ability to produce clean energy.
"By using computation, we can understand the properties and the behavior of this molecule and gain insight into improving it," says Cheung. "If we can capture the mechanism that converts solar energy into chemical fuel, it opens the door to many opportunities."
The results of this study were published in the Journal of Physical Chemistry B in February 2012.
Testing the Triad
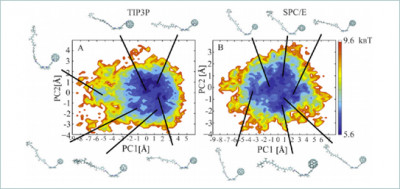
This image shows the 2-D free energy of the system as a function of the first principal component (PC1) and the second principal component (PC2) for the triad in the bulk system. The color scale is given in units of kBT, where kB is the Boltzmann constant and T is temperature at 350 K. Six representative structures of the triad are illustrated in each panel. Image credit: Margaret Cheung.
Unlike typical photovoltaic cells, which are made out of solid-state materials, the carotenoid-porphyrin-C60 molecular triad is a bioinorganic compound, combining biological and inorganic components. These hybrid molecules are more flexible, fragile and prone to breaking.
The light-harvesting molecular triad that Cheung investigates combines three components: a carotenoid (an organic pigment, similar to the chromophore in plants); a fullerene or buckyball (a carbon-based molecule that forms a hollow sphere); and a porphyrin (an organic compound that can bind ligands to metals, as with hemoglobin).
"When photons hit the triad, the molecule becomes excited," Cheung explained. "This excited state scatters the electrons, providing a driving force to move electrons into a polarized distribution, like a dipole."
This separation of positive and negative charges in the system becomes the stored chemical potential from which energy can be produced. The problem is that bioinorganic compounds are flexible in nature and cannot remain in a fixed configuration over a short period of time. "We want to harness this charge-separated state, but if the vehicle that carries the charge-separated state is wobbling all the time, then it's not very reliable," Cheung said.
The wobbliness of the triad also challenged efforts to simulate its dynamics in the past. Cheung had to pioneer new methods that combine quantum chemistry approaches, molecular dynamics simulations, and statistical physics to take into account the microscopic landscape of the molecules and the many configurations that the triad might be in when photons hit the material.
Cheung and her team simulated the triad in solution at many different temperatures and confinement conditions to map the impact of these changes on the behavior of the molecule. They discovered that the triad conformation distribution could be manipulated by temperature fluctuations in the solvent. Furthermore, they concluded that when the presence of confinement is considered, the network of solvent molecules is disrupted, which dictates both the positions of the components in the confinement and the triad's attraction to the wall.
Ultimately, the goal is to use information from the computer simulations to design a scalable system that maximizes the generation of chemical energy while maintaining the triad's stability.
"If we want to stabilize the triad, there are many different strategies to do so," Cheung said. "Maybe this involves redesigning the molecules. Maybe it involves the design of different solvents or different capsule sizes. We don't know unless we try different conditions and probe its responses. By using computation, we can understand the properties and the behavior of this molecule, which can give us some insight into how to improve them."
“NERSC was a great resource for us,” adds Cheung. “The systems ran so efficiently that we were able to get our results in a reasonable time-frame, which made us more effective.”
This story was adapted from the feature “Biologically Inspired Energy” written by Aaron Dubrow of TACC.
About NERSC and Berkeley Lab
The National Energy Research Scientific Computing Center (NERSC) is a U.S. Department of Energy Office of Science User Facility that serves as the primary high performance computing center for scientific research sponsored by the Office of Science. Located at Lawrence Berkeley National Laboratory, NERSC serves almost 10,000 scientists at national laboratories and universities researching a wide range of problems in climate, fusion energy, materials science, physics, chemistry, computational biology, and other disciplines. Berkeley Lab is a DOE national laboratory located in Berkeley, California. It conducts unclassified scientific research and is managed by the University of California for the U.S. Department of Energy. »Learn more about computing sciences at Berkeley Lab.